|
NO2 is a covalent compound because the bond is formed between one nitrogen and two oxygen atom by the sharing of electrons. Iron (III) Chloride is an ionic compound, its formula unit is FeCl3. the number of charges on an ion formed by a non-metal is equal to the group number minus eight. 
the number of charges on an ion formed by a metal is equal to the group number of the metal.There is a quick way to work out what the charge on an ion should be: Therefore, the iron cation in iron(II) chloride has a charge of 2+, and the charge on the iron cation in iron(III) chloride has a charge of 3+. What is the charge on the cation in the ionic compound FeCl2? There are two Cl- ions in the compound, so each Cl must have oxidation number -1. The total of the oxidation numbers of all the atoms or ions in a neutral compound is defined as zero. In FeCl2, the Fe2+ ion has oxidation number +2. This system is used only for elements that form more than one common positive ion….Naming Ions. Thus, Fe2+ is called the iron(II) ion, while Fe3+ is called the iron(III) ion. Iron(III) chloride/IUPAC ID What charge does Fe have in fe en 3 2+? 7 Which is the correct name for FeCl3 compound?Īlso called ferric chloride, it is a common compound of iron in the +3 oxidation state. 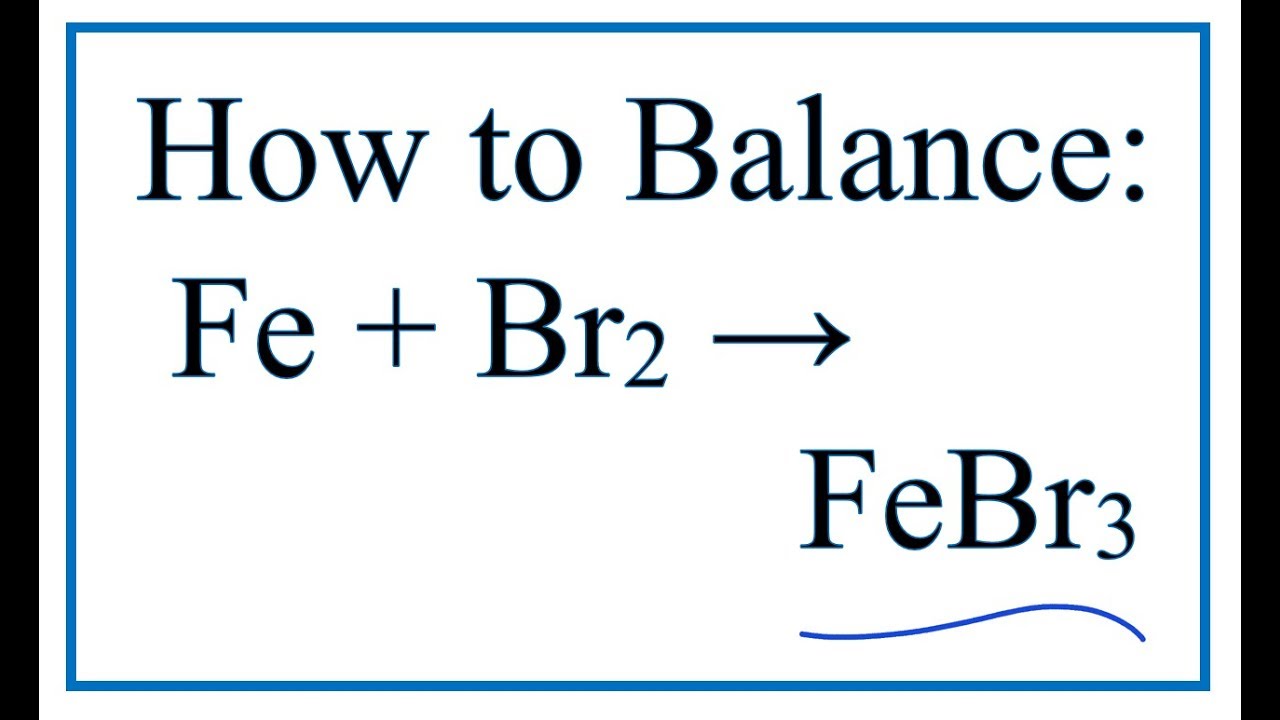
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
